IASR 42(7), 2021【THE TOPIC OF THIS MONTH】COVID-19 in Japan as at May 2021
The topic of This Month Vol.42 No.7(No. 497)
COVID-19 in Japan as at May 2021
(IASR Vol. 42 p135-136: July 2021)
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative virus of coronavirus disease 2019 (COVID-19), is classified into the family Coronaviridae, genus Betacoronavirus, and has a positive-sense, single-stranded RNA genome consisting of approximately 30,000 bases. SARS-CoV-2 uses angiotensin-converting enzyme 2 (ACE2) as a receptor to attach to and enter human cells. SARS-CoV-2 is an enveloped virus and is inactivated by alcohol, detergents, etc.Global and domestic epidemic trends
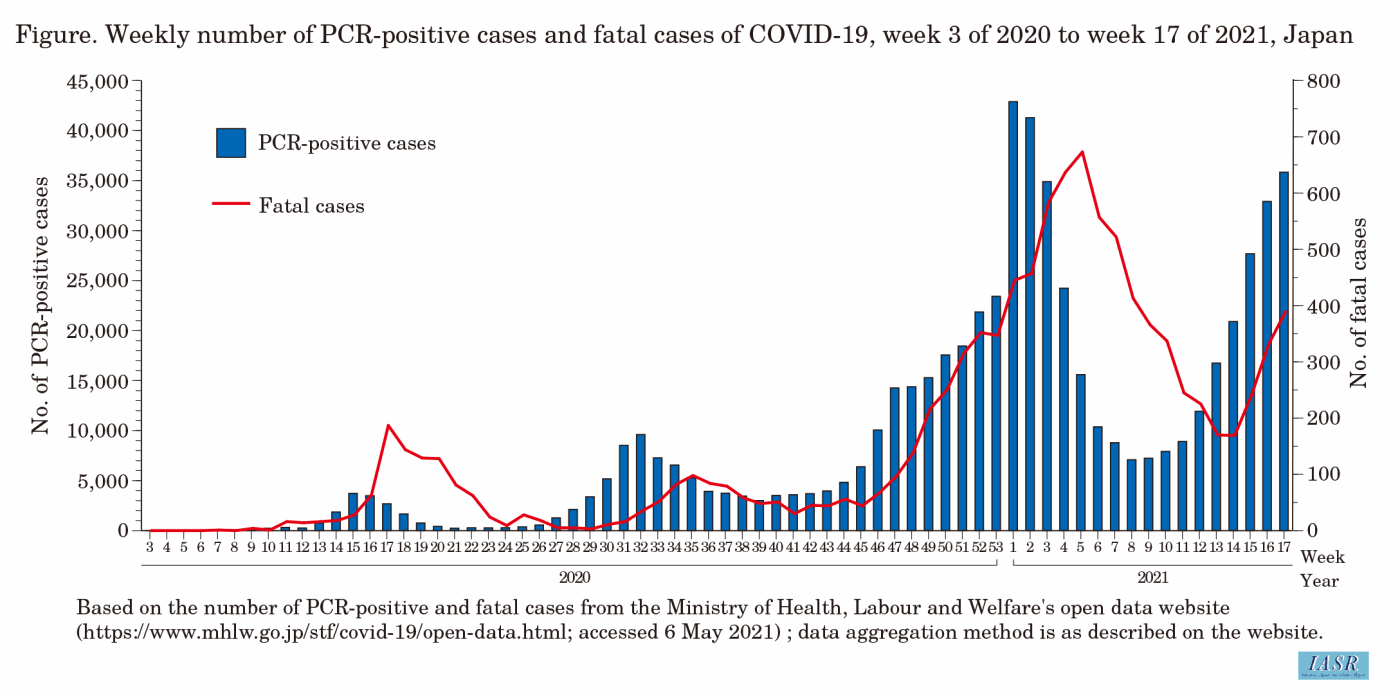
COVID-19, which first appeared in Wuhan, China, in December 2019, spread worldwide in a short period of time and a pandemic state was declared by the World Health Organization (WHO) on March 11, 2020. According to the WHO COVID-19 Weekly Epidemiological Update (as at May 30, 2021) published on June 1, 2021, the cumulative number of reported cases was 169,604,858 and the cumulative number of reported deaths was 3,530,837 (https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---1-june-2021).According to the open data provided by the Ministry of Health, Labour and Welfare (MHLW) in Japan (https://www.mhlw.go.jp/stf/covid-19/open-data.html), since the first patient was confirmed on January 16, 2020, 598,298 PCR-positive cases and 10,358 deaths have been reported through May 2, 2021 (week 17 of 2021) (Figure). There have been recurrent COVID-19 epidemics since April 2020; the number of PCR-positive cases and patients requiring inpatient treatment began to increase again in mid-March 2021, and the number of severe cases also began to increase in late March, overwhelming the medical care provision system. As a result, the government declared a semi-state of emergency in 10 prefectures, including Tokyo and Osaka, on April 5, 2021. On April 25, as a more stringent policy, the government declared a state of emergency in 4 prefectures, including Tokyo and Osaka, followed by 6 other prefectures, including Aichi, Hokkaido, and Okinawa.
According to the domestic epidemiological surveillance data of COVID-19 (preliminary figures as at 18:00 on May 26, 2021) (https://www.mhlw.go.jp/content/10906000/000785178.pdf) released by the MHLW, the male/female ratio of the PCR-positive cases was 1.2 and the proportionate distribution of the PCR-positive cases by age group was, in order of the highest to lowest proportion, those in their 20s (22.2%), followed by those in their 30s (14.9%), 40s (14.4%), 50s (13.1%), 60s (8.5%), 70s (7.5%), 80s and over (7.4%), 10s (7.3%), and under 10 years (3.2%) (excluding 1.4% of those with age information unknown, under investigation, and/or undisclosed). The number of reported cases is high for the 20s and 30s, even when considering the population size for each age group. The proportion of fatal cases among the reported PCR-positive cases by age group was as follows: 80 years or older (13.2%), 70s (4.8%), 60s (1.3%), 50s (0.3%), 40s (0.1%), and those in their 30s or younger (0.0%). Those aged 80 years or older accounted for 7.4% of PCR-positive cases but made up 64.7% of all fatal cases (rounded to the first decimal place).
Genomic surveillance and emergence of variants of concern with increased transmissibility and/or changes in antigenicity
In Japan, genomic surveillance has been carried out since the emergence of COVID-19 (https://www.niid.go.jp/niid/ja/basic-science/467-genome/9586-genome-2020-1.html). The National Institute of Infectious Diseases (NIID), public health institutes (PHIs), quarantine stations, and others are cooperating to perform viral genome analysis (see p.137 & 139 of this issue) and the analyzed data are used for cluster investigations and monitoring purposes, together with epidemiological information (see p.141 of this issue).Since the end of 2020, the emergence of SARS-CoV-2 variants with mutations that can affect the transmissibility, severity, antigenicity, etc., has become a problem. In particular, the B.1.1.7 lineage (alpha variant) first detected in the UK, the B.1.351 lineage (beta variant) first detected in South Africa, the P.1 lineage (gamma variant) first detected in Japan in a traveler returning from Brazil, and the B.1.617.2 lineage (delta variant) first detected in India are of global concern. As a countermeasure against these variants, genome surveillance was strengthened in Japan. Virus genome analysis is attempted for all imported PCR-positive cases detected at quarantine, and, within the country, PHIs perform the nucleic acid detection test (PCR) that can detect the N501Y mutation that is seen in the B.1.1.7 lineage (alpha variant), the B.1.351 lineage (beta variant), and the P.1 lineage (gamma variant), and viral genome analysis is performed at PHIs or NIID for screen-positive cases.
In Japan, B.1.1.7 (alpha variant) was imported from overseas at the end of 2020 with sporadic clusters of cases, which prompted investigation (see p.137 of this issue). However, the reported number and proportion of the B.1.1.7 lineage (alpha variant) that has the N501Y mutation increased nationwide.
Regarding the emergence of new variants, in addition to the assessment of transmissibility, severity, antigenicity, etc., and phenotypes, it is necessary to examine whether the currently used nucleic acid detection test (e.g.,PCR) can detect these variants (see p.143 of this issue).
Treatment and vaccines
As a treatment against COVID-19, remdesivir and dexamethasone have been approved and used in Japan depending on the severity, etc. On April 23, 2021, baricitinib [Janus kinase (JAK) inhibitor] was also approved (Clinical Guidance for COVID-19, 5th edition). For cases with a severity of moderate II or above, heparin, an anticoagulant, is used in combination with the above drugs. In addition, there are many drugs that are under development against COVID-19 both domestically and overseas.The development and rollout of the COVID-19 vaccine have progressed rapidly, and several vaccines have shown to have high eff icacy and good safety in randomized controlled trials. As at May 2021, vaccines manufactured by Pfizer, Moderna, and AstraZeneca have been approved in Japan. According to the website of the Prime Minister of Japan and His Cabinet (https://www.kantei.go.jp/jp/headline/kansensho/vaccine.html), 3.14 million healthcare workers and 0.47 million elderly individuals (total of 3.61 million individuals; approximately 2.9% of the population in Japan) have been vaccinated twice as at June 1, 2021. Continuous monitoring of vaccine effectiveness and safety is essential, and monitoring of adverse events has started together with the initial rollout of the vaccines in the country (see p.145 of this issue).
Future challenges
In some countries where vaccine rollout started early, public health and social measures are starting to relax. Under these current circumstances, the following are the challenges related to COVID-19.First is the evaluation of the duration of vaccine effectiveness. The follow-up period of randomized controlled trials evaluating the eff icacy of vaccines that have already been used is as short as about 2 months after the second dose and the study subjects have already been unblinded. It is necessary to evaluate vaccine effectiveness over time, which can be influenced by waning immunity and the relaxation of public health and social measures.
Next is the development and evaluation of COVID-19 vaccines that are in clinical trials and preclinical trials in Japan and overseas. Estimation of the level of immunity required for protection from infection and symptom development, based on the level of immunity (humoral immunity, cell-mediated immunity, etc. ) attained by the vaccines currently in use, is being considered as one of the criteria to evaluate effectiveness.
The WHO, together with the National Institute for Biological Standards and Control in the United Kingdom, has created and distributed the International Standard for anti-SARS-CoV-2 immunoglobulin. It is desired to standardize the antibody measurement system in each clinical trial using this product (see p.147 of this issue). With the possibility of needing booster vaccines in the medium to long term, the continuous development of safe and effective vaccines will reduce vaccine hesitancy. With multiple effective vaccines, we may be better equipped to combat newly emergent variants and may be able to narrow the vaccine gap between developed and developing countries, which has been a noted concern.
The final issue is the emergence of variants with altered antigenicity. To this end, it is necessary to reduce the opportunities for the virus to mutate by suppressing the circulation of the virus and by facilitating early detection and response through viral genome surveillance. With regards to quarantine, continuous preparedness, monitoring, and response to prevent or delay the importation of variants from overseas are essential. In addition, in cases of reinfection and breakthrough infection (infection after vaccination), a virus that escapes immunity may be selected, and it is necessary to monitor such cases and investigate the cause of these infections.
In the face of these issues, while reducing the number of susceptible individuals with vaccines, the continuation of public health and social measures, such as avoiding the so-called “three C’s”, maintaining physical distance, wearing masks, hand hygiene, etc., are advised, and continuous compliance will be required.

