IASR 42(8), 2021【THE TOPIC OF THIS MONTH】Vancomycin-resistant enterococcus (VRE) infection in Japan
Published: 26 December 2024
PDF download(PDF:463KB)
The topic of This Month Vol.42 No.8(No. 498)
Vancomycin-resistant enterococcus (VRE) infection in Japan
(IASR Vol. 42 p155-156: August 2024)
Enterococcus is a gram-positive coccus that resides in the intestinal tract and the environment. It is isolated from stool cultures of healthy individuals and may also contaminate urine samples. Examples of the enterococcal species involved in human infections include E. faecalis, E. faecium, E. gallinarum, and E. casseliflavus, and approximately 70% of the enterococci isolated from clinical specimens are E. faecalis. Enterococci are opportunistic pathogens that cause infections, such as bacteremia, endocarditis, and urinary tract and abdomen/pelvis infections in immunocompromised hosts (e.g., older adults; those with diabetes, malignant tumors, or heart disease; and post-operative patients). Bacteremia and endocarditis are particularly serious, and bacteremia caused by E. faecium has a high case-fatality rate. Vancomycin is an extremely important antibacterial drug for infections caused by enterococci that are naturally resistant to cephems, carbapenems, and aminoglycosides.
Vancomycin-resistant enterococci (VRE) infection has been a notifiable disease since April 1999, based on the Infectious Diseases Control Law. The Infectious Diseases Control Law defines VRE infection as an infectious disease caused by enterococci resistant to vancomycin. Notification is required when VRE with MIC of ≥ 16 µg/mL is isolated in specimens that should normally be sterile (e.g., blood, ascites, pleural effusion, and cerebrospinal fluid), or when isolated in specimens that are not normally sterile (e.g., sputum, pus, and urine) and the isolate is determined to be the causative organism of the infectious disease (for the notification criteria, see: https://www.mhlw.go.jp/bunya/kenkou/kekkaku-kansenshou11/01-05-14-01.html).
Mechanisms of vancomycin resistance (see p.157 of this issue)
Glycopeptides, such as vancomycin and teicoplanin, are cell wall synthesis inhibitors. Resistance is caused by a decreased aff inity of glycopeptides to the bacterial cell wall. VanA, VanB, VanD, and VanM are resistant types that are known to confer high resistance. VanA is generally highly resistant to both vancomycin and teicoplanin, whereas VanB is highly resistant to vancomycin but susceptible to teicoplanin. On the other hand, VanC exhibits low resistance to vancomycin and is susceptible to teicoplanin, and thus, resistance to glycopeptides due to VanC is rarely a clinical problem.
National Epidemiological Surveillance of Infectious Diseases (NESID)
When VRE infection became a notifiable disease in 1999, the criteria for reporting VRE were that the vancomycin MIC of the isolated enterococci be ≥16 µg/mL, or the detection of resistance genes vanA, vanB, or vanC. However, E. gallinarum and E. casseliflavus naturally possess vanC on their chromosomes, and since VRE only with VanC rarely poses a clinical problem, the requirement of detecting a vancomycin resistance gene was removed in April 2013, and since then, the drug susceptibility test has been the sole criteria. As a result of this change, among those reported cases through 2012, namely E. faecium, E. casseliflavus, E. gallinarum, and E. faecalis, notified cases of E. casseliflavus and E. gallinarum markedly decreased since 2013 and E. faecium has accounted for the majority (>80%) of notifications in recent years (Figure).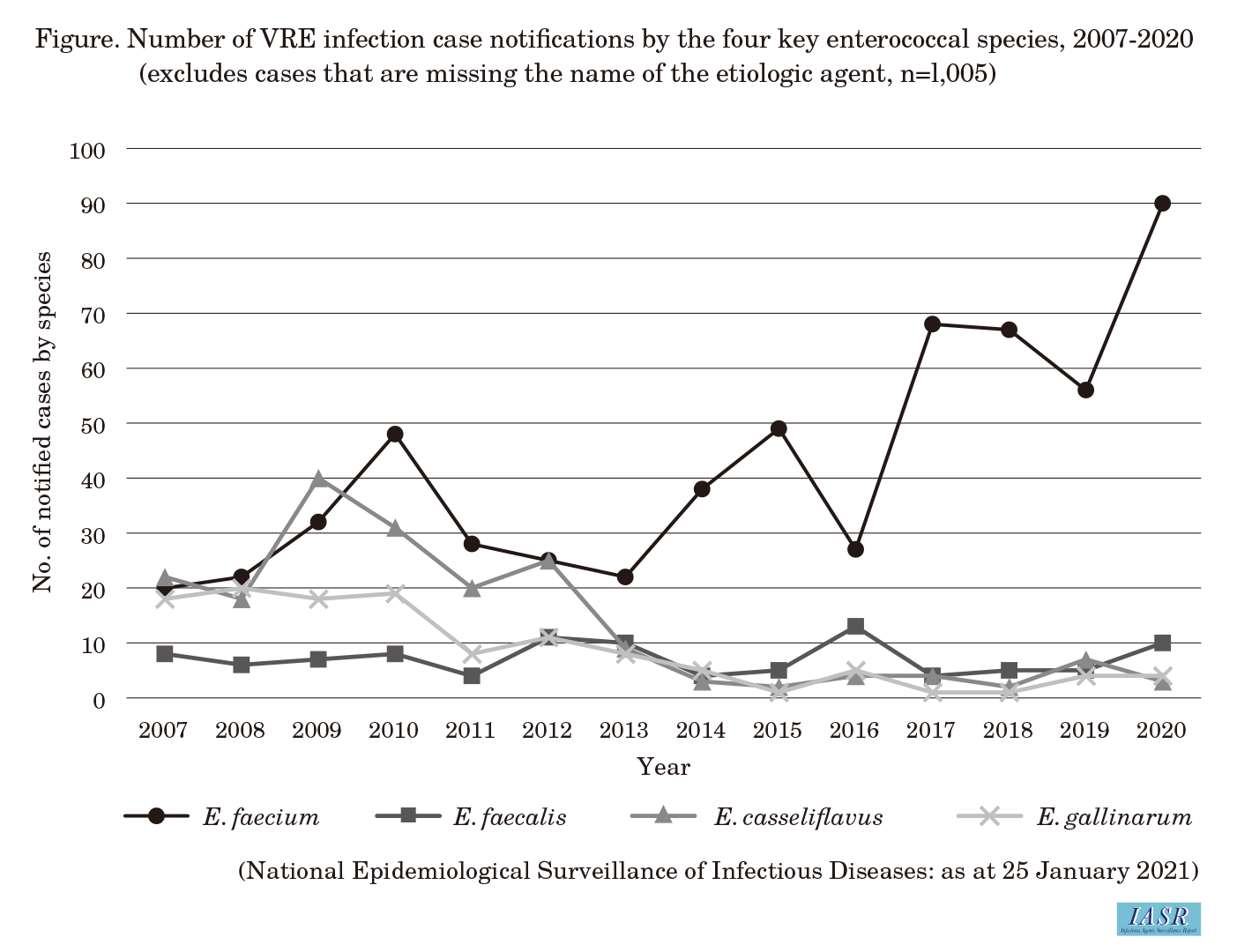
The number of notified cases through NESID remained below 100 per year from 2011-2019. However, there were 135 cases in 2020, which exceeded the previous peak of 120 cases in 2010. The number of prefectures with notifications also increased from 15 in 2013, when the number of notifications was the lowest at 55 in the past ten years, to 26 in 2020, an approximately 1.7-fold increase (IASR 42: 100-101, 2021). Based on changes in the reported bacterial species, most of these increases are assumed to be due to vancomycin-resistant E. faecium.
A 2017 notice (Health Service Bureau / Tuberculosis and Infectious Diseases Control Division Notification 0328 No. 4) stated that testing should be encouraged at regional public health institutes (PHIs) and aff iliate laboratories when a VRE infection is reported. As a result, systems to carry out testing at PHIs are being strengthened (see p.158 of this issue). In terms of the response to VRE infection outbreaks in medical facilities, a 2014 notice (Health Policy Bureau / Regional Medical Care Planning Division Notification 1219 No. 1) stated that, in line with recognition of an outbreak, strict infection prevention and control measures must be implemented after the detection of the first case (including a case with colonization). VRE outbreaks occurred primarily in relatively small medical facilities with long-term care beds until the early 2000s but later increased in acute care hospitals, and multiple large-scale nosocomial infection events occurred in core acute care hospitals between 2018-2019 (IASR 42: 100-101, 2021 and see p.160 of this issue). It is important that medical facilities cooperate with public health centers and regional PHIs from an early stage to prevent the outbreak from becoming larger (see p.162 of this issue).
Ampicillin is used to treat penicillin-sensitive VRE infections, whereas daptomycin or linezolid is the main treatment for ampicillin-resistant VRE infections (see p.163 of this issue).
Trends in VRE infections overseas
The isolation of VRE from humans was first reported in Europe in 1988 and then in the United States (U.S.) in 1989. Since then, VRE has spread rapidly in medical facilities in the U.S. The spread of VRE in U.S. medical facilities has been attributed to the increased use of vancomycin in medical facilities. Through the 1990s, VRE infections were reported mainly in the U.S. and Europe, where advanced medical care is prevalent, but since then, VREs have spread to almost all regions of the world. Since the late 2010s, the VRE isolation rate has increased in multiple regions overseas, which is suggested to be due to the emergence of new epidemic strains (see p.165 of this issue).
VRE in imported meat
In some European and Asian countries, the long-term use of veterinary avoparcin, a glycopeptide antimicrobial agent similar to vancomycin, led to increased VRE in livestock and its transmission to humans. Many VanA-type VRE have also been detected in imported meat in Japan. The use of avoparcin was banned in many countries by around 2000, and the frequency of VRE detection in imported meat has been decreasing. However, VanN-type VRE has been continuously isolated from domestic chicken meat, and its link to imported chicks has been suspected (see p.166 of this issue). Therefore, ongoing monitoring is deemed necessary.
Conclusions
The number of reported VRE infections in Japan is extremely low compared to other countries, and VRE remains a rare antimicrobial-resistant pathogen. According to the Ministry of Health, Labour and Welfare’s Japan Nosocomial Infections Surveillance program (JANIS), VRE has been isolated in only 10% of the participating medical facilities. However, in the context of an increasing VRE isolation rate in multiple regions overseas, multiple cases of large-scale nosocomial infection events have also been reported in Japan in recent years, mainly in core acute care hospitals. Large-scale nosocomial infection outbreaks in core medical facilities not only affect the local medical system (e.g., restrictions on new admissions) but also spread VRE to small- and medium-sized medical facilities through patient transfers. This increases the risk of spreading VRE throughout the medical network and beyond.
Surveillance conducted under the NESID targets VRE infection in symptomatic patients, but once a nosocomial infection is confirmed, active screening of patients, including carriers, is required for an extended period of time. This results in large economic losses due to the temporary closure of the medical facility and the high costs associated with infection prevention and control, including screening. Therefore, when a VRE infection is notified, the isolated strain must be obtained and sent to a PHI to ensure laboratory testing, considering the possibility of regional spread. Furthermore, it is desired that medical facilities and local governments share medically-relevant VRE infection surveillance information with each other, and public health centers and PHIs take the lead in infection prevention and control measures.

